Our missions
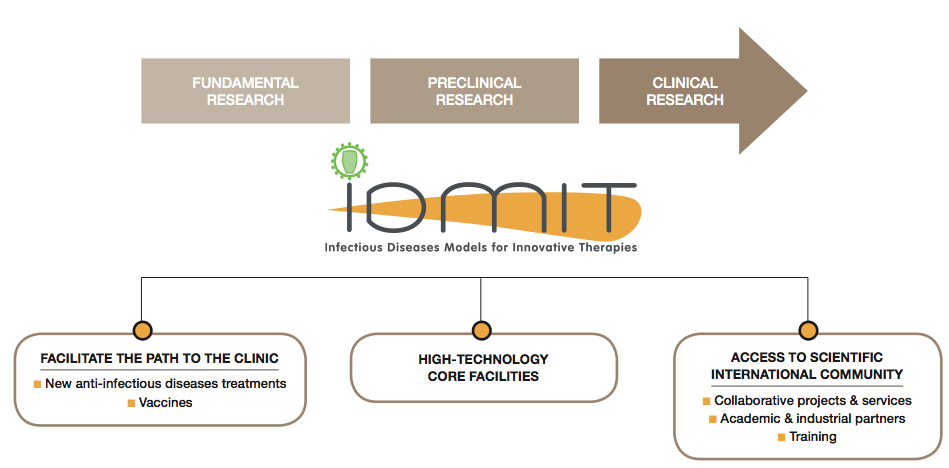
IDMIT is a Research and Technological Center/Department, providing the scientific community with exceptional resources for:
- developing and characterizing relevant infected non-human primate models,
- studying and understanding host/infectious diseases relationships,
- evaluating innovative preventive/prophylactic and/or therapeutic strategies.
The co-founders partners, all international leaders in the field of research into host pathogen interactions, provide complementary expertise with outstanding core facilities for monitoring diseases, treatments and prevention in relevant animal models of human infections. Tools already developed by the partners are extended with state-of-the-art equipments and standardized assays.
IDMIT is also fostering research programs for the development of new models of human infections and for refinement of the use of animals and reduction of the number of animals required for experimental preclinical researches. There are already and will be new programs on assays standardization and harmonization, non invasive methods (telemetry, in vivo imaging, aerosol administration), animal training and in silico modeling. A Biological Resource Center has been created in partnership with our industrial partner Oncodesign Services.
The major objective of this consortium is to provide the national and international scientific community with a highly competitive infrastructure for preclinical research, facilitating the discovery of markers of safety and efficacy for new vaccines and treatments and accelerating the translation of innovations at the bench into clinical practice.
IDMIT provides to external partners, including public academic institutions and private companies, with access to its core facilities, for the achievement of their R&D projects, the testing of new products (vaccine, innovative therapeutics…), the development of prototypes and the performance of pilot studies. This is done in accordance with quality assurance requirements for future industrialization and release onto the market. Quality criteria [ISO9001 (2015 version) obtained in July 2017, and renewed in 2020] is met with the assistance of Oncodesign services, a company specialized in assay standardization and quality insurance.
Finally, IDMIT is an attractive site for training and teaching in the fields of “preclinical research” and “technological developments”, through the contribution of its founder partners, including the Institut Pasteur and Paris-Saclay University, INSERM, which are highly reputed teaching institutions.
IDMIT develops new non-human primate (NHP) preclinical models and monitors core facilities, equipments and transversal tools to evaluate:
– new vaccines,
– new therapies,
– microbicides,
– adjuvants of immunity,
– innovative delivery approaches.
against several human pathogens:
– HIV/AIDS,
– Alphaviruses,
– Flaviviruses,
– Coronaviruses,
– Influenza/Flu,
– Tuberculosis,
– Human STDs (Sexually Transmitted Diseases),
– Malaria,
– Chlamydia,
– Respiratory pathogens,
– Pathogens of bioterrorism.